|
1/13/2024 0 Comments No2 molecular geometry 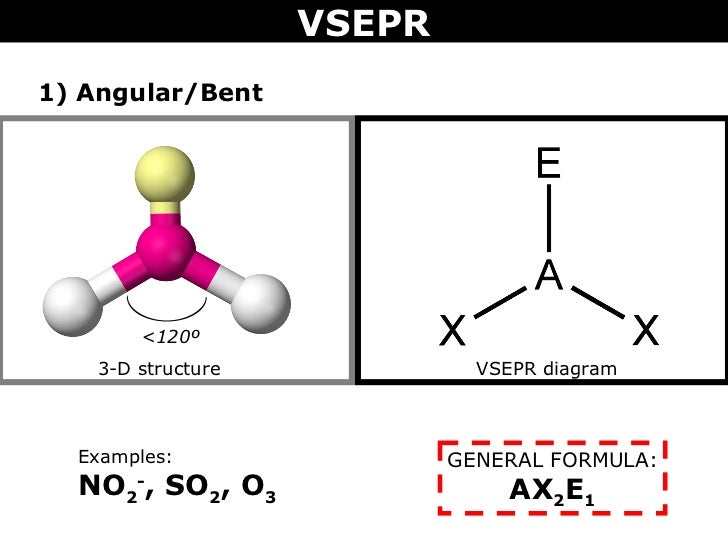
The actual bond angle for nitrogen dioxide is By comparison, the angle found in the nitrite ion, or #NO_2^(-)#, is because now nitrogen has attached a full lone pair of electrons that experience greater repulsion with the oxygen atoms. The molecule's geometry will still be closer to bent, but the bond angle will be larger than the ideal such a geometry predicts, mostly because the oxygen atoms experience less repulsion from the single electron present on the nitrogen atom. Nitrogen Dioxide (NO 2) is one of a group of highly reactive gases known as oxides of. The simple way to determine the hybridization of NO2 is by counting the bonds and lone electron. Thus NO2 adopts a shape or molecular geometry different from its ideal electron pair geometry, i.e., bent or V-shaped. However, it is due to the presence of a lone pair of electrons on the central N-atom that lone pair-bond pair repulsions exist in the molecular ion. In this case, the nitrogen dioxide molecule is considered to have a #AX_2E_0.5# geometry, which is right between #AX_2E_0# - linear, and #AX_2E_1# - bent. The molecular geometry of SO2 is bent, with a bond angle of 120. Nitrogen Dioxide (NO2) involves an sp2 hybridization type. The electron geometry of the NO2 ion is trigonal planar. Now, VSEPR Theory can be extended to accomodate odd-electron molecules. More specifically, nitrogen dioxide has a total of 17 valence electrons - 5 from nitrogen and 6 from each of the two oxygen atoms that comprise the molecule. the number of valence electrons the molecule has is not an even number. Sketch and name the three different shapes that this molecule might have. 9 A molecule with the formula AB 2, in which A and B represent different atoms, could have one of three different shapes. Nitrogen dioxide, or #NO_2#, is an odd-electron molecule, i.e. SO 3 2 Answer a Answer b Answer c Answer d Click here to see a video of the solution PROBLEM 5.2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
